 Group - electronegativity decreases as you go down a group. Elements on the right side of the period table only need a few electrons to complete the octet, so they have strong desire to grab another atom's electrons. As a result, they have low electronegativity. Why? Elements on the left of the period table have 1 -2 valence electrons and would rather give those few valence electrons away (to achieve the octet in a lower energy level) than grab another atom's electrons. Period- electronegativity increases as you go from left to right across a period. Why? The shielding affect makes it easier to remove the outer most electrons from those atoms that have many electrons (those near the bottom of the chart).Įlectronegativity is an atom's 'desire' to grab another atom's electrons Group - ionization energy decreases as you go down a group. Elements on the left of the chart would prefer to give up their electrons so it is easy to remove them, requiring less energy (low ionization energy). The means it will require more energy to remove the outer most electron. Why? Elements on the right of the chart want to take others atom's electron (not given them up) because they are close to achieving the octet. Period - ionization energy increases as you go from left to right across a period. It is closely related to electronegativity 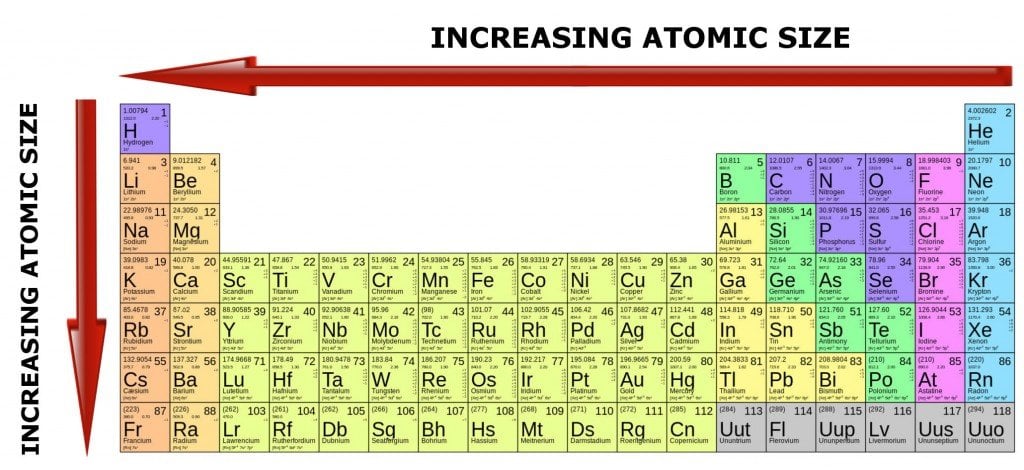
Ionization energy is the amount of energy required to remove the outmost electron. Additionally, new energy levels of elections clouds are added to the atom as you move from period to period down a group, making the each atom significantly more massive, both is mass and volume. Why? There is a significant jump in the size of the nucleus (protons + neutrons) each time you move from period to period down a group. 
Group - atomic radius increases as you go down a group. Why? Stronger attractive forces in atoms (as you go from left to right) between the opposite charges in the nucleus and electron cloud cause the atom to be 'sucked' together a little tighter. Period - atomic radius decreases as you go from left to right across a period. Similarly, as we proceed across the row, the increasing nuclear charge is not effectively neutralized by the electrons being added to the 2 s and 2 p orbitals.Summary of Periodic Table Trends Atomic Radius Consequently, beryllium is significantly smaller than lithium. 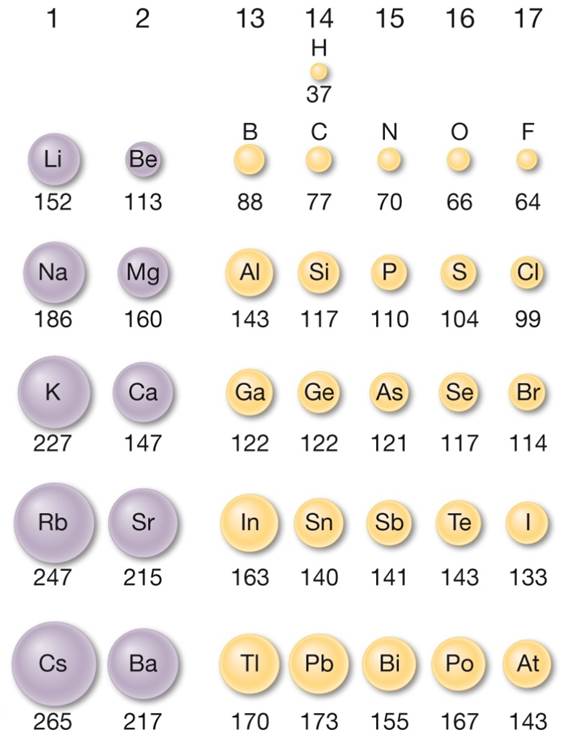
This means that the effective nuclear charge experienced by the 2 s electrons in beryllium is between +1 and +2 (the calculated value is +1.66). (More detailed calculations give a value of Z eff = +1.26 for Li.) In contrast, the two 2 s electrons in beryllium do not shield each other very well, although the filled 1 s 2 shell effectively neutralizes two of the four positive charges in the nucleus. Thus the single 2 s electron in lithium experiences an effective nuclear charge of approximately +1 because the electrons in the filled 1 s 2 shell effectively neutralize two of the three positive charges in the nucleus. Although electrons are being added to the 2 s and 2 p orbitals, electrons in the same principal shell are not very effective at shielding one another from the nuclear charge. All have a filled 1 s 2 inner shell, but as we go from left to right across the row, the nuclear charge increases from +3 to +10. The atoms in the second row of the periodic table (Li through Ne) illustrate the effect of electron shielding. The greater the effective nuclear charge, the more strongly the outermost electrons are attracted to the nucleus and the smaller the atomic radius.Ītomic radii decrease from left to right across a row and increase from top to bottom down a column. 
For all elements except H, the effective nuclear charge is always less than the actual nuclear charge because of shielding effects. \( \newcommand\)) experienced by electrons in the outermost orbitals of the elements. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
